COVID-19
This page is for adult patients. For pediatric patients, see: COVID-19 (peds)
. For pregnant patients see COVID-19 in pregnancy.
Background
- Caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
- See COVID-19: Epidemiology and pathophysiology
Clinical Features
Initial Presentation
- Many patients are asymptomatic
- At onset of symptoms: fever, dry cough, myalgias, fatigue, shortness of breath
- Fever and cough start early, shortness of breath noted about 9 days into illness
- Fever not present in all adults
- Only 1/2 of patients may have fever at time of admission[1]
- less common in vulnerable populations
- Less common: cough with sputum, sore throat, headache, congestion, GI symptoms, anosmia, altered mental status
- Sudden onset anosmia has a high specificity for COVID-19 infection
| Symptom[2] | % |
| Fever | 87.9 |
| Dry cough | 67.7 |
| Fatigue | 38.1 |
| Sputum production | 33.4 |
| Shortness of breath | 18.6 |
| Myalgia or arthralgia | 14.8 |
| Sore throat | 13.9 |
| Headache | 13.6 |
| Chills | 11.4 |
| Nausea or vomiting | 5.0 |
| Nasal congestion | 4.8 |
| Diarrhea | 3.7 |
| Hemoptysis | 0.9 |
| Conjunctivitis | 0.8 |
Common Complications
The initial presentation can be followed by delayed and serious complications
- Pulmonary
- Most common complications: pneumonia, ARDS (average 8 days from onset, 20% of patients in China)
- "Happy Hypoxemia": many of these patients will be hypoxic without dyspnea
- Decompensation risk occurs during 2nd week of illness leading to respiratory failure
- Cardiac Complications[3]
- Myocarditis,Acute Myocardial infarction, Dysrhythmias, cardiomyopathy, venous thromboembolism.
- Vasodilatory shock reported in 67% of ICU admissions
- Cardiomyopathy reported in 33% of ICU admissions
- Mortality reported 67% of ICU admissions
- Myocarditis,Acute Myocardial infarction, Dysrhythmias, cardiomyopathy, venous thromboembolism.
- Neurological Complications- thought to be related to the increased levels of interleukin (IL)-6, IL-12, IL-15, and tumor necrosis factor alpha (TNF-α)[4]
- acute CVA, encephalitis, Guillain-Barré syndrome, acute necrotizing hemorrhagic encephalopathy, and hemophagocytic lymphohistiocytosis
Differential Diagnosis
Influenza-Like Illness
- Influenza
- Parainfluenza
- URI
- Pneumonia
- Sinusitis
- Toxic exposure
- Pyelonephritis
- Bronchitis
- Coronavirus
Causes of Pneumonia
Bacteria
Viral
- Common
- Influenza
- Respiratory syncytial virus
- Parainfluenza
- Rarer
- Adenovirus
- Metapneumovirus
- Severe acute respiratory syndrome (SARS)
- Middle east respiratory syndrome coronavirus (MERS)
- 2019-nCoV (COVID-19)
- Cause other diseases, but sometimes cause pneumonia
Fungal
- Histoplasmosis
- Coccidioidomycosis
- Blastomycosis
- Pneumocystis jirovecii pneumonia (PCP)
- Sporotrichosis
- Cryptococcosis
- Aspergillosis
- Candidiasis
Parasitic
Evaluation
Workup
Consider minimal to no workup in well-appearing patients with mild disease
Viral Testing
- RT-PCR (reverse transcriptase polymerase chain reaction) is most commonly used test for confirming cases
- Sensitivity may be only 75%, but highly specific
- Turnaround time may be several hours to days
- Real time RT-PCR e.g. Cepheid
- Rapid test with results in <1hr
- Serologic testing for IgM/IgG is not widely available, but likely more sensitive
- The presence of IgG with a negative RT-PCR likely confirms past exposure and some immunity
- Test kit availability varies widely by region and institution
Labs
Consider in sicker patients (likely requiring admission):
- Chemistry
- CBC w/diff
- Coagulation studies
- PT/PTT/INR - DIC possible
- D-dimer, fibrinogen - markers of severity
- LFTs - mild elevation of ALT/AST
- Inflammatory Markers
- Troponin [9] - myocarditis
- Sepsis labs
- Lactate
- Blood culture x2
- Swabs - Co-infection has been reported as high as 7-20%
- Flu swab
- Respiratory viral panel
- Note that BIOFIRE Respiratory Panel Corona Virus assay does NOT detect this COVID-19 subtype
- Urine pregnancy test in reproductive-age women
- Other labs to consider in patients that will be admitted:
- HBV serologies, HCV antibody
- Consider (as clinically indicated): PCP DFA, beta-d-glucan, urine legionella Ag, IL-6
Imaging
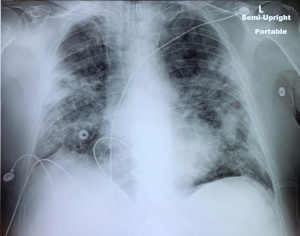
X-ray
- Portable CXR preferred in PUI to prevent spread of infection
- May be normal in early disease
- Typical pattern is peripheral patchy ground glass opacities (GGO)
- More opacities correlates with worse disease
- GGOs may coalesce and appear as infiltrates
- Not every PUI needs a chest X-ray. Patients who are more likely to need one include any moderate or high acuity patient, elderly, concerning chronic conditions, BMI > 40, high risk socioeconomic situations.
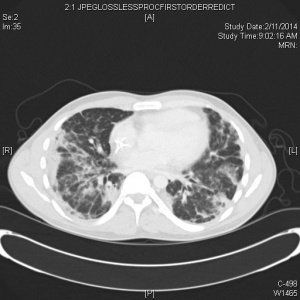
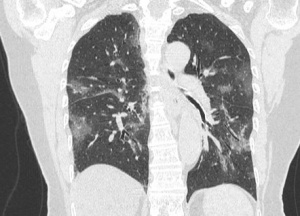
CT
- Many have normal imaging early on (CDC does not recommend CT for diagnostic purposes)
- CT (86%) more sensitive than CXR (59%) for detecting GGOs
- Generally, the findings on chest imaging are not specific and overlap with other infections, including influenza, H1N1, SARS and MERS.[10]
US
- Uncertain role in diagnosis at this time
- May reveal B lines, consolidation, or "ragged" appearance of pleural line
- Useful in evaluating undifferentiated Dyspnea
Diagnosis
- Typically confirmed by viral testing (see above)
Disease Severity
Some define moderate and severe acuity as follows
- Low: SaO2 > 93% on RA, RR < 20, and HR < 110
- Moderate: SaO2 = 91-93% on RA, RR 20-24, HR 110-124 with wheezing, rales, or an otherwise abnormal lung exam.
- High: SaO2 < 91%, RR > 24, HR > 124.
^ If febrile, treat with acetaminophen and reassess acuity.
Prediction of Need for Intubation
- ROX Index for intubation after high-flow nasal cannula (HFNC}
- Predicts HFNC failure/need for intubation
- https://www.mdcalc.com/rox-index-intubation-hfnc
Management
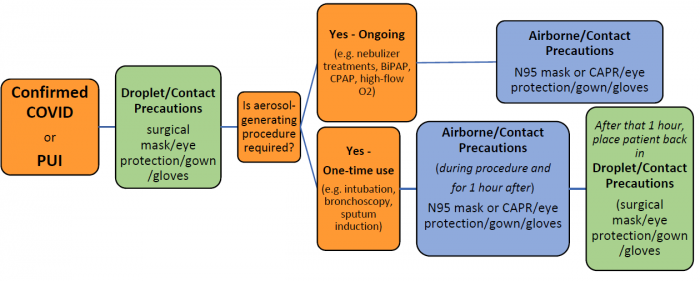
COVID-19 PPE Summary Table
| Contact Category | Precations | Room Type |
| General (all persons) | Social distancing; meticulous hygiene; basic mask | NA |
| Undifferentiated patients at risk (e.g. prior to evaluation or testing) | Contact and droplet precautions, including eye protection | Negative-pressure NOT required |
| Persons Under Investigation | Contact and droplet precautions, including eye protection | Negative-pressure NOT required |
| Aerosol-Generating Procedures | Contact and airborne precautions, including eye protection | Negative-pressure required |
See prevention of COVID-19 transmission in the healthcare setting for full PPE recommendations
General Supportive Care
Pulmonary [11]
- Supplemental oxygen therapy if Sat<90%
- Target SPO2 92%-96%
- High-flow Nasal Cannula
- Some guidelines recommend HFNC over BIPAP/CPAP, in those that fail low-flow O2. [12]
- Requires patient to be on airborne isolation.
- Non-Invasive Ventilation if no HFNC
- Consider awake proning to improve oxygenation
- Bronchodilators if bronchospasm present
- Use metered-dose inhaler (avoid nebulizers due to aerosolization)
Other
- Infectious disease
- Acetaminophen for fever
- Consider antibiotics for bacterial pneumonia coverage
- Cardiovascular [13]
- Most patients without hemodynamic compromise
- Maintain euvolemia - hypervolemia may contribute to ARDS
- Hypoperfusion - cautious fluid resuscitation
- Vasopressors
- 1st line - Norepinepherine (alternative: epinephrine)
- 2nd line - Vasopressin
- Vascular
- Consider anticoagulation
Medications by Patient Category


Outpatients Not Requiring Admission[16]
- High risk patients (one of the following, if available):
- Ritonavir-boosted nirmatrelvir (Paxlovid)
- Sotrovimab
- Remdesivir
- Molnupiravir
Dexamethasone has not demonstrated benefit in this patient category and may be potentially harmful
Outpatients Requiring Home Oxygen (New or Increased)[17]
- Dexamethasone 6mg PO daily x the duration of supplemental oxygen (not to exceed 10 days)
- Consider Remdesivir
Hospitalized Not Requiring Oxygen
- Consider Remdesivir
Dexamethasone has not demonstrated benefit in this patient category and may be potentially harmful
Hospitalized Requiring Oxygen
- Dexamethasone 6mg PO daily x 10 days
- Remdesivir 200 mg IV once, then 100 mg IV once daily for 4 days or until hospital discharge (whichever comes first).
- If requiring High-Flow or NIV, add baricitinib or IV tocilizumab
- If not available, IV sarilumab can be used
Respiratory failure
Intubation of Potential COVID-19 Patients
Aerosol-generating procedure: see this link for PPE recommendations and related precautions
- Use checklist if available (see example: File:Harbor COVID Airway Management v3-16-20.pdf)
- Use BVM with viral filter or avoid BVM altogether, if possible
- Use RSI to prevent coughing gagging; consider higher dosing of paralytics.
- Use video laryngoscopy to keep provider face further away from patient (afterwards, clean with grey wipes, observe 3 min wet time)
Lung Protective Mechanical Ventilation
Lung Protective Ventilator Settings[18] should be the default for all intubated patients, unless contraindicated. It has demonstrated mortality benefit for ARDS-like pulmonary conditions; limits barotrauma and decreases complications of high FiO2[19][20]
- Mode
- Volume-assist control
- Tidal Volume
- Start 6-8cc/kg predicted body weight[21]
- Predicted/"ideal" body weight is used because a person's lung parenchyma does not increase in size as the person gains more weight.
- Titrate down if plateau pressure >30 mmHg
- Start 6-8cc/kg predicted body weight[21]
- Inspiratory Flow Rate (comfort)
- More comfortable if higher rather than lower
- Start at 60-80 LPM
- Respiratory Rate (titrate for ventilation)
- Average patient on ventilator requires 120mL/kg/min for eucapnia
- Start 16-18 breaths/min
- Maintain pH = 7.30-7.45
- FiO2/PEEP (titrate for oxygenation)
- Move in tandem to achieve:
- SpO2 BETWEEN 88-95%
- PaO2 BETWEEN 55-80mmHg
- See also deterioration after intubation
COVID Lung Phenotypes and Their Management
Hypoxemic patients can be divided into two general phenotypes[22]
COVID L Lung Phenotype
- Characterized by Low elastance (i.e., high compliance), Low ventilation to perfusion ratio, Low lung weight and Low recruitability
- Often referred to as the “happy hypoxemic”
- Normal lung volumes and low lung recruitability.
- Hypoxemia may be due to loss of regulation of perfusion and loss of hypoxic vasoconstriction.
- These patients can be damaged iatrogenically if you respond to their pulse ox with standard vent modes
- Do poorly with low tidal volume (TV) and high PEEPs
- Best managed with high FiO2 which allows you to limit the PEEP
- Recommended initial vent settings:
- 8 ml/kg TV, 100% FiO2
- Increase the PEEP only if the patient is desaturating on a high FiO2.
- Can turn into COVID H patients on the vent.
COVID H Lung Phenotype
- Characterized by High elastance, High right-to-left shunt, High lung weight and High recruitability.
- Increased permeability of the lung leads to edema, atelectasis, decreased gas volume, and decreased TV for a given inspiratory pressure.
- High degree of lung recruitability.
- 20 – 30% of patients fit ARDS criteria:
- Hypoxemia
- Bilateral infiltrates
- Decreased the respiratory system compliance
- Increased lung weight and potential for recruitment
- The ARDS ladder applies only to this subset of COVID patients.
Contraindicated Therapies
- NSAIDS
- There is anecdotal evidence to suggest that NSAIDs could potentially harm patients infected with COVID-19.[23]
- Some experts suggest avoiding NSAIDs altogether while recommending the use of paracetamol/acetaminophen instead. [24]
- It is important to note that there is no strong evidence to suggest NSAIDs should be avoided in general in COVID-19 patients[25]
- ACEi/ARBs
- There is an increase in mortality in patients with both hypertension and COVID-19 infection. [26]
- ACEi and ARBs, used in the treatment of hypertension, has been postulated to contribute to the increased mortality by upregulating membrane-bound angiotensin-converting enzyme 2 (ACE2) which allows COVID-19 entry into human cells. [27]
- Currently, however, there is insufficient evidence to recommend against using ACEi and ARBs in patients with COVID-19. [28]
- Nebulizers
- Avoid nebulizers as they are generally ineffective and may aerosolize virus
- Albuterol with spacer is safer, though probably ineffective unless co-occuring reactive airway disease
- MDI equivalents: Albuterol or ipratropium
- <20 kg or 5yrs old: 4-5 puffs with a spacer every 20 minutes. 4 breaths between puffs.
- >20 kg or 5yrs old: 8-10 puffs with a spacer every 20 minutes. 4 breaths between puffs.
- MDI equivalents: Albuterol or ipratropium
Disposition
- 80% of patients do not require hospital admission
- Mild cases for persons under investigation for Covid-19 awaiting a positive test result can self quarantine at home in conjunction with the local Public Health Dept
- "Silent hypoxemia" is now reported in patients with oxygen saturations ranging in the 80s-90s without respiratory distress. Hypoxia is not recommended as an absolute indication for emergent intubation.
- Note: symptoms may worsen over 2nd week of illness
Admission
- Hospitalize: Respiratory distress/failure, multi-organ failure, rapid disease progression requiring escalating supportive care. Meets criteria for high acuity above. Moderate acuity with extra risk factors (pneumonia, immunosuppressed, elderly, comorbidities), complicated social situation, worsening symptoms > 10 days out.
- PSI/PORT, MuLBSTA, and CURB65 scores have all been proposed criteria for admission and predicting outcomes.
- These scores are not externally validated. Use with caution. https://www.mdcalc.com/covid-19#calcs
- PSI/PORT, MuLBSTA, and CURB65 scores have all been proposed criteria for admission and predicting outcomes.
- May consider discontinuation of hospital isolation when:
- Resolution of fever without anti-pyretic, resolution of symptoms, and negative COVID19 testing
Special Situations
- For pregnant patients see: COVID-19 in pregnancy
- For pediatric patients see: COVID-19 in pediatrics
COVID-19 and STEMI
- According to ACC consensus statement "During the COVID-19 pandemic, PCI remains the standard of care for STEMI patients"
- If thrombolytics are indicated options include:
- Administer 10u Retavase (reteplase) IV bolus followed by a second bolus at 30 minute rather than PCI. OR
- Tenecteplase (TNKase) 30 mg IV bolus
- If Tenecteplase is not available, it is acceptable to administer a lower dose of alteplase (tPA) at 50 mg (8 mg bolus, followed by 42 mg infusion over 90 minutes).
- Follow thrombolytics by 40u/kg heparin (max dose 4,000 units) IV and 600mg clopidogrel PO and ASA 325 mg PO
COVID-19 and CPR
- Interim AHA Guidance
- Don all PPE prior to initiating CPR. CPR is aerosol generating.
- Intubate early, video laryngoscopy preferred
- Pause chest compressions during intubation
- If patient is on ventilator at time of arrest consider leaving patient on ventilator
- Adjust ventilator to allow for asynchronous ventilation
- If using BVM then attach high efficiency particulate air (HEPA) filter
- Use of mechanical compression device (e.g. LUCAS) is encouraged
- Use auto CPR device if available
Prognosis
COVID-19 Risk Factors for Severe Disease [29]
See VACO calculator
See Also
External Links
- https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/hospitalized-adults--therapeutic-management/
- WHO COVID-19 Situation Dashboard (Live): https://experience.arcgis.com/experience/685d0ace521648f8a5beeeee1b9125cd
- CDC Main Healthcare Page
- EMCrit Covid Airway Management
- Johns Hopkins COVID Case Map (Live):https://coronavirus.jhu.edu/map.html
- California Emergency Medical Services Authority Resource Portal https://emsa.ca.gov/covid19/
- Oxford Journal of Travel Medicine COVID19 Resources https://academic.oup.com/journals/pages/coronavirus?cc=gb&lang=en&
- EM:RAP Corependium COVID-19 (open access) https://www.emrap.org/corependium/chapter/rec906m1mD6SRH9np/Novel-Coronavirus-2019-COVID-19?MainSearch=%22covid%22&SearchType=%22text%22
- MedRxiv Covid-19 SARS
- Additional DPH Guidance: http://publichealth.lacounty.gov/acd/nCorona2019.htm
- Seattle ICU doctor's one page info on mgmt of COVID from ACEP website File:COVID19 seattle one pager.pdf
- Harbor DEM COVID airway management guide File:Harbor COVID Airway Management v3-16-20.pdf
- Proper donning and doffing from CDC File:Doffing PPE CDC.pdf
- Radiopaedia COVID-19 Resources (https://radiopaedia.org/articles/covid-19)
- Literature review https://reacting.inserm.fr/literature-review/
- COVID related trials: https://covid.inato.com/analysis
References
- ↑ 1. Zhou F, Yu T, Du R et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet. 2020;395(10229):1054-1062. doi:10.1016/s0140-6736(20)30566-3
- ↑ World Health Organization. "Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19)" (PDF): 11–12. Retrieved 5 March 2020.
- ↑ Long B, Brady WJ, Koyfman A, Gottlieb M. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020;38(7):1504-1507. doi:10.1016/j.ajem.2020.04.048
- ↑ Bridwell R, Long B, Gottlieb M. Neurologic complications of COVID-19. Am J Emerg Med. 2020;38(7):1549.e3-1549.e7. doi:10.1016/j.ajem.2020.05.024
- ↑ Yang X, Yu Y, Xu J et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. The Lancet Respiratory Medicine. 2020. doi:10.1016/s2213-2600(20)30079-5
- ↑ Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020. doi:10.1007/s00134-020-05991-x
- ↑ Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020. doi:10.1007/s00134-020-05991-x
- ↑ Young B, Ong S, Kalimuddin S et al. Epidemiologic Features and Clinical Course of Patients Infected With SARS-CoV-2 in Singapore. JAMA. 2020. doi:10.1001/jama.2020.3204
- ↑ https://www.covidprotocols.org
- ↑ From the American College of Radiology (3/11/20):
- ↑ Alhazzani W, Møller M, Arabi Y et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med. 2020. doi:10.1007/s00134-020-06022-5
- ↑ Alhazzani W, Møller M, Arabi Y et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med. 2020. doi:10.1007/s00134-020-06022-5
- ↑ Alhazzani W, Møller M, Arabi Y et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med. 2020. doi:10.1007/s00134-020-06022-5
- ↑ https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/clinical-management-summary/
- ↑ https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/clinical-management-summary/
- ↑ https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/clinical-management-summary/
- ↑ https://www.covid19treatmentguidelines.nih.gov/management/clinical-management/clinical-management-summary/
- ↑ The Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med. 2000;342(18):1301-1308.
- ↑ ARDSnet
- ↑ O'Brien J. Absorption Atelectasis: Incidence and Clinical Implications. AANA Journal. June 2013. Vol. 81, No. 3.
- ↑ Brower RG, et al. "Ventilation With Lower Tidal Volumes As Compared With Traditional Tidal Volumes For Acute Lung Injury And The Acute Respiratory Distress Syndrome". The New England Journal of Medicine. 2000. 342(18):1301-1308.
- ↑ Gattinoni L et al. Covid-19 pneumonia: different respiratory treatment for different phenotypes. Intensive Care Medicine. 2020. https://www.esicm.org/wp-content/uploads/2020/04/684_author-proof.pdf
- ↑ Willsher, Kim. “Anti-Inflammatories May Aggravate Covid-19, France Advises.” The Guardian, Guardian News and Media, 14 Mar. 2020, www.theguardian.com/world/2020/mar/14/anti-inflammatory-drugs-may-aggravate-coronavirus-infection.
- ↑ Day, Michael. “Covid-19: Ibuprofen Should Not Be Used for Managing Symptoms, Say Doctors and Scientists.” Bmj, 2020, p. m1086., doi:10.1136/bmj.m1086.
- ↑ Pergolizzi JV Jr, Varrassi G, Magnusson P, et al. COVID-19 and NSAIDS: A Narrative Review of Knowns and Unknowns. Pain Ther. 2020;9(2):353-358. doi:10.1007/s40122-020-00173-5
- ↑ Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. Published online March 13, 2020. doi:10.1001/jamainternmed.2020.0994
- ↑ Fang, Lei, et al. “Are Patients with Hypertension and Diabetes Mellitus at Increased Risk for COVID-19 Infection?” The Lancet Respiratory Medicine, vol. 8, no. 4, 2020, doi:10.1016/s2213-2600(20)30116-8.
- ↑ Patel AB, Verma A. COVID-19 and Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers: What Is the Evidence? JAMA. Published online March 24, 2020. doi:10.1001/jama.2020.4812
- ↑ Massachusetts General Hospital COVID-19 Treatment Guide Version 1.36 04/05/2020. https://www.massgeneral.org/assets/MGH/pdf/news/coronavirus/mass-general-COVID-19-treatment-guidance.pdf. Published 2020. Accessed April 8, 2020.