Vulvovaginitis
(Redirected from Vaginal discharge)
Background
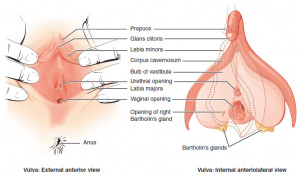
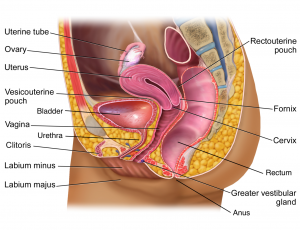
- Definition: inflammation of the vulva and vagina
- Candida Vaginitis and Contact Vulvovaginitis may occur in pre-pubertal and non-sexually active people
- Atrophic vaginitis may occur in postmenopausal women
Vulvovaginitis
- Bacterial vaginosis
- Candida vaginitis
- Trichomonas vaginalis
- Contact vulvovaginitis
- Bubble baths and soaps
- Deodorants, powders, and douches
- Clothing
- Atrophic vaginitis due to lack of estrogen (AKA Vulvovaginal atrophy)
- Lichen sclerosus
- Tinea cruris
- Chlamydia/Gonorrhea infection
- Pinworms
- Vaginal foreign body
- Toilet paper
- Other
- Genitourinary syndrome of menopause
- Foreign body
- Allergic reaction
- Normal physiologic discharge
Clinical Features
- Nonspecific
- Discharge
- Itching
- Erythema or rash
- Dysuria
- Odor
- Pain with intercourse
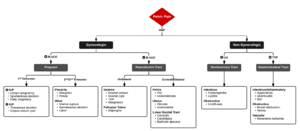
Differential Diagnosis
Acute Pelvic Pain
Gynecologic/Obstetric
- Normal variants may be noted on exam but generally do not cause pain or other symptoms
- Nabothian cysts: Epithelial cells within mucous glans that appear as yellow inclusions on the cervix
- Cervical Ectropion: Edothelial cells on the exterior of the cervix
- Parous cervix: The is no longer round but may have multiple shapes after birth
- Pregnancy-related
- Ectopic pregnancy
- Spontaneous abortion, threatened or incomplete
- Septic abortion
- Pelvic organ prolapse
- Acute Infections
- Vulvovaginitis
- Adnexal Disorders
- Hemorrhage/rupture of ovarian cyst
- Ovarian torsion
- Twisted paraovarian cyst
- Other
- Myoma (degenerating)
- Genitourinary trauma
- Ovarian hyperstimulation syndrome
- Sexual assault
- Recurrent
- Mittelschmerz
- Primary/Secondary Dysmenorrhea
- Pelvic Congestion Syndrome
- Endometriosis
Genitourinary
Gastrointestinal
- Gastroenteritis
- Appendicitis
- Bowel obstruction
- Perirectal abscess
- Diverticulitis
- Inflammatory bowel disease
- Irritable bowel syndrome
- Mesenteric adenitis
Musculoskeletal
- Abdominal wall hematoma
- Psoas hematoma, psoas abscess
- Hernia
Vascular
- Pelvic thrombophlebitis
- Abdominal aortic aneurysm
- Ischemic bowel (Mesenteric Ischemia)
Evaluation
Workup
- Upreg
- Evaluate for infectious causes with cultures, wet mount, and trichomonas NAAT (or molecular vaginitis panel)
- Respiratory or enteric flora may be seen in vaginal cultures from pre-puberal females
- Respiratory: Staph, Strep, H influenza, Moraxella, and N meningitidis
- Enteric: E coli, proteus, klebsiella, shigella etc.
- STIs are less common
- Candida and gardnerella may be see on wet mount
- Respiratory or enteric flora may be seen in vaginal cultures from pre-puberal females
- Consider O&P
- Pinworms may cause vulvovaginitis in children [2]
Diagnosis
Management
Empiric Treatment
- Acceptable when clinically suspecting BV, vaginal candidiasis, or a non-infectious etiology
- If signs/symptoms are equivocal for a specific diagnosis or if they suggest possible trichomoniasis, diagnostic testing should first be performed prior to treatment.
Non-Infectious Etiology Treatment
- Hygienic measures for non-specific vulvovaginitis particularly in pre-pubertal females
- OTC Vaginal lubrication products for post-menopausal women with vulvovaginal atrophy symptoms [3]
- Consider hormone replacement therapy in post-menopausal women
- Best provided by a primary care provider
Treatment for Test-Positive Patients based on Organism and Symptomatology
| Positive Organism | Symptomatic | Asymptomatic | Test of cure needed? | Treat sexual partner(s)? |
|---|---|---|---|---|
| BV | Treat | Do not treat | No, unless persistent symptoms | No |
| Vaginal candidiasis | Treat | Do not treat | No, unless persistent symptoms | No |
| Trichomoniasis | Treat | Treat | Yes (in 3 months; use NAAT) | Yes |
Bacterial vaginosis
First Line Therapy[4]
- Metronidazole 500 mg PO Twice Daily for 7 days OR
- Metronidazole gel 0.75%, one full applicator (5 g) intravaginally, Daily for 5 days OR
- Clindamycin cream 2%, one full applicator (5 g) intravaginally Nightly for 7 days
Metronidazole does not cause a disulfiram-like reaction with alcohol.[5]
Alternative Regimin
- Clindamycin 300 mg PO BID for 7 days OR
- Clindamycin ovules 100 mg intravaginally qHS for 3 days (do not use if patient has used latex condom in last 72 hours)
Other regimens have been studied and have varying efficacy compared to placebo but due to cost and availability do not represent alternatives outside of absolute contraindications to preferred regimens.
Pregnant
- Metronidazole 500mg PO Twice a day x 7 days[4]
- Metronidazole 250mg PO Three times a day has also been studied[6][7]
- Although metronidazole crosses the placenta, no evidence of teratogenicity or mutagenic effects among infants has been reported in multiple cross-sectional, case-control, and cohort studies of pregnant women[4]
Prophylaxis (Sexual Assault)
- Metronidazole 500mg PO Twice a day x 7 days[8]
Candida vulvovaginitis
Uncomplicated
There is little resistance to azole medications; treatment often dictated by patient preference.
- Fluconazole 150mg PO once[9]
- Intravaginal therapy
- Clotrimazole 1% cream applied vaginally for 7 days OR 2% applied vaginally for 3 days
- Miconazole 2% cream applied vaginally for 7 days OR 4% cream x 3 days
- Butoconazole 2% applied vaginally x 3 days
- Tioconazole 6.5% applied vaginally x 1
Complicated
Severe or immunosuppressed
- Fluconazole 150mg PO q72h x 3 doses
Non-albicans species
- For example, C. glabrata, C. krusei and other atypical Candida spp.
- Boric acid vaginal suppository intravaginal qday x ≥14 days
- Can be fatal if taken orally
- If empirically treated and later is found to have non-albicans Candida spp., no change in therapy is needed if patient is improving (otherwise switch to boric acid.
Recurrent (≥ 4 infections in a year)
- Treat as for uncomplicated (see above)
- Once therapy completed, prescribe long-term treatment
- Fluconazole 150mg PO qweek x 6 months OR
- Intravaginal medication, such as clotrimazole 500mg PV qweek or 200mg PV twice a week
Pregnant Patients
- Intravaginal clotrimazole or miconazole are the only recommended treatments
- Duration is 7 days
- PO fluconazole associated with congenital malformations and spontaneous abortions[10]
[[Trichomonas vaginalis
Non-Pregnant
- Metronidazole 500mg PO BID for 7 days [11]
- Tinidazole 2g PO once
Pregnant
Only treat if the patient is symptomatic
- Metronidazole 500mg PO BID for 7 days [12]
Sexual Partner Treatment
- Female: Same as above
- Male: Metronidazole 2 gm PO x1 [13]
Disposition
- Virtually all patients can be discharges with PCP or Gyn follow up
- Any test sent should be followed for results
See Also
External Links
- ACOG patient information for vaginitis [6]
References
- ↑ Norris DL, Young JD. UTI. EM Clin N Am. 2008; 26:413-30.
- ↑ Loveless M, Myint O. Vulvovaginitis- presentation of more common problems in pediatric and adolescent gynecology. Best Pract Res Clin Obstet Gynaecol. 2018 Apr;48:14-27. doi: 10.1016/j.bpobgyn.2017.08.014. Epub 2017 Sep 5. PMID: 28927766.
- ↑ Alvisi S, Gava G, Orsili I, Giacomelli G, Baldassarre M, Seracchioli R, Meriggiola MC. Vaginal Health in Menopausal Women. Medicina (Kaunas). 2019 Sep 20;55(10):615. doi: 10.3390/medicina55100615. PMID: 31547180; PMCID: PMC6843679.
- ↑ 4.0 4.1 4.2 CDC Sexually Transmitted Infections Treatment Guidelines, 2021.[1]
- ↑ Is combining metronidazole and alcohol really hazardous?[2]
- ↑ Reduced incidence of preterm delivery with metronidazole and erythromycin in women with bacterial vaginosis[3]
- ↑ Effect of metronidazole in patients with preterm birth in preceding pregnancy and bacterial vaginosis: a placebo-controlled, double-blind study[4]
- ↑ Sexual Assault and Abuse and STIs – Adolescents and Adults[5]
- ↑ Pappas PG, Kauffman CA, Andes DR, et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 2016; 62:e1.
- ↑ Molgaard-Nielsen D et al. Association Between Use of Oral Fluconazole During Pregnancy and Risk of Spontaneous Abortion and Stillbirth. JAMA. 2016;315(1):58-67.
- ↑ Workowski KA, Bachmann LH, Chan PA, et al. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm Rep 2021;70(No. RR-4):1–187. DOI: http://dx.doi.org/10.15585/mmwr.rr7004a1external icon
- ↑ CDC Trichomoniasis 2021. https://www.cdc.gov/std/treatment-guidelines/trichomoniasis.htm
- ↑ Workowski KA, Bachmann LH, Chan PA, et al. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm Rep 2021;70(No. RR-4):1–187. DOI: http://dx.doi.org/10.15585/mmwr.rr7004a1external icon